

THORSTEN HOPPE
University of Cologne
CECAD Cluster of Excellence
INSTITUTE FOR GENETICS
CECAD Research Center
Joseph-Stelzmann-Str. 26
50931 Cologne
Tel.: 0049(0)221478 - 84216
Tel.: 0049(0)221478 - 84210
Fax.: 0049(0)221478 - 84217
office-hoppe(at)uni-koeln.de
Public record at ORCID
Proteostasis in Development and Aging
Accumulation of damaged proteins is associated with age-related neurodegeneration in Alzheimer’s and Parkinson’s patients. The maintenance of protein homeostasis, or proteostasis, involves the degradation of misfolded and damaged proteins, and is essential for cellular function, organismal growth, and ultimately viability. Sustaining proteostasis is not only a long-term challenge for individual cells but also for entire organisms, since damaged proteins accumulate with stress and aging. Not all tissues are equally susceptible to the toxicity of protein aggregates, suggesting tissue-specific differences in proteostasis pathways.
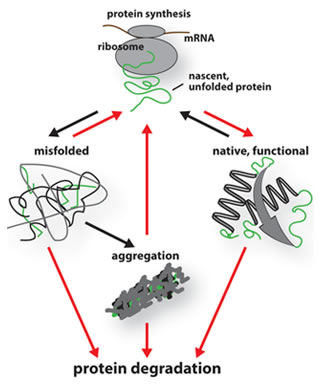
The maintenance of protein homeostasis, or proteostasis, involves folding and degradation of damaged proteins. It is commonly thought that age-related impairment of protein quality control (PQC) affects general proteostasis networks, causing enhanced aggregation of misfolded proteins that can be toxic for cells and shortens organismal lifespan.
The ubiquitin/proteasome system (UPS) is a major proteolytic route functioning in a cellular network that maintains the proteome during stress and aging. Another proteolytic system supporting proteostasis is the autophagy-lysosome pathway that degrades proteins inside activated autophagosomes. An age-related impairment of either of these systems causes enhanced protein aggregation and affects lifespan, suggesting functional overlap and cooperation between UPS and autophagy in stress and aging. The ultimate goal of our research is to assemble a global picture of stress-induced proteolytic networks critical for aging of multicellular organisms.
Selected Publications
Original Papers
Efstathiou S., Ottens F., Schütter L.S., Ravanelli S., Charmpilas N., Gutschmidt A., Le Pen J., Gehring N.H., Miska E.A., Bouças J., Hoppe T. (2022). ER-associated RNA silencing promotes ER quality control. Nat. Cell Biol. 12, 1714-1725.
Segref A., Vakkayil K.L., Padvitski T., Li Q., Kroef V., Lormann J., Körner L., Finger F., Hoppe T. (2022). Thermosensation in Caenorhabditis elegans is linked to ubiquitin-dependent protein turnover via insulin and calcineurin signalling. Nat Commun. 13, 5874.
Finger F., Ottens F., Springhorn A., Drexel T., Proksch L., Metz S., Cochella L., Hoppe T. (2019). Olfaction regulates organismal proteostasis and longevity via microRNA-dependent signaling. Nature Metabolism 1, 350–59.
Tawo R., Pokrzywa W., Kevei E., Akyuz M.E., Balaji V., Arian S., Höhfeld J., Hoppe T. (2017). The Ubiquitin Ligase CHIP Integrates Proteostasis and Aging by Regulation of Insulin Receptor Turnover. Cell 169, 470-82.
Kuhlbrodt K., Janiesch P.C., Kevei E., Segref A., Barikbin R., and Hoppe T. (2011). The Machado-Joseph disease deubiquitylase ATX-3 couples longevity and proteostasis. Nat. Cell Biol. 13, 273-81.
Ottens F., Franz A., and Hoppe T. (2021). Build-UPS and break-downs: metabolism impacts on proteostasis and aging. Cell Death Differ. doi: 10.1038/s41418-020-00682-y.
Hoppe T. and Cohen E. (2020). Organismal protein homeostasis mechanisms. Genetics. 215, 889-901.
Höhfeld J. and Hoppe T. (2018). Ub and down: ubiquitin exercise for the elderly. Trends Cell Biol. 28, 512-522.
Tissue-specific proteostasis:
Proteostasis Networks in Muscle Development and Maintenance
The correct folding and assembly of proteins and protein complexes is essential for cellular function. Eukaryotes evolved quality control pathways, including chaperones and the ubiquitin/proteasome system (UPS), which correct or eliminate protein damage to maintain a healthy proteome. Intensive cooperation between protein folding and degradation pathways ensures cellular protein homeostasis (proteostasis) and prevents the accumulation of aggregated proteins and the manifestation of neurodegenerative diseases.
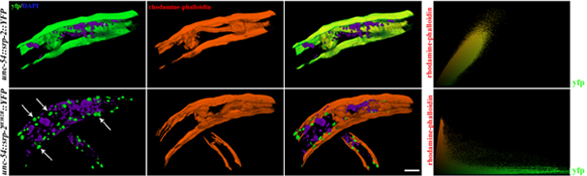
The assembly and maintenance of myofilaments requires a tightly balanced proteostasis network. One key player important for muscle thick filament formation in health and disease is the myosin assembly chaperone UNC-45 (Janiesch et al., Nat Cell Biol., 2007; Gazda et al., Cell, 2013). However, little is known about how folding and degradation networks are adjusted to maintain cellular proteostasis during the lifetime of multicellular organisms. Therefore, our long-term objective is to understand how the balance between protein folding and degradation is coordinated with muscle assembly and its maintenance. To this end, we combine genetic and biochemical approaches to study the conserved function of UNC-45 in myosin assembly and examine how this function is modulated under stress conditions and during the aging process. Specifically, we use targeted screening strategies to uncover chaperones, UPS and autophagy components that are required for muscle function. The identified effector networks will be studied with regard to modulation of myosin folding and assembly under different physiological conditions in C. elegans and C2C12 mouse myoblasts. Finally, we would like to address remodeling of proteostasis network interactions during aging.
A combination of genetic, biochemical and in vivo imaging techniques allows us to examine the dynamic cross talk between folding and degradation networks by focusing on the prominent muscle specific chaperone UNC-45. Given that the organization and arrangement of sarcomere proteins are highly conserved in C. elegans, we expect that our research will have broad implications for the understanding of myosin assembly, human myopathies and proteostasis mechanisms in general.
Organismal proteostasis:
Cell-Non-Autonomous Control of Ubiquitin-Dependent Protein Turnover
The ubiquitin/proteasome system (UPS) mediates turnover of damaged proteins supporting maintenance of the cellular proteome, organismal health, and lifespan. We recently developed an in vivo degradation assay based on a fluorescently labelled model substrate that allows monitoring ubiquitin-dependent protein turnover in different tissues of C. elegans (Segref et al., Genetics, 2011) We used this assay truly successfully in different genetic approaches and identified novel key players regulating the UPS in response to proteotoxic conditions including oxidative stress, DNA damage, and aging. Intriguingly, we observed similar proteolytic defects for human metabolic diseases such as for example isovaleric acidemia caused by human IVD mutations (Segref et al., Cell Metab., 2014), thus providing an evolutionarily conserved link for metabolic pathways vital for protein turnover both in worms and humans.
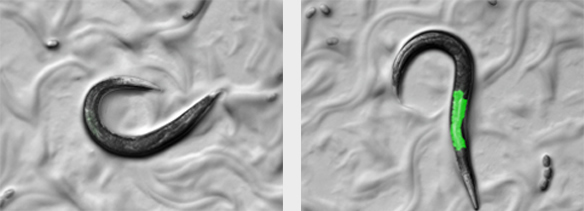
quantification of protein turnover in different tissues of C. elegans. The right panel shows a mutant worm with proteolytic defects and substrate stabilization.
Until now much of our knowledge regarding ubiquitin-mediated protein degradation evolved from single-celled studies such as yeast or tissue culture cells providing a deep mechanistic understanding of substrate recognition, ubiquitylation, and proteasomal turnover. However, the physiological relevance of the UPS in organismal proteostasis, its regulation by intercellular communication, and response mechanisms induced upon environmental cues remain fascinating questions. In fact, we recently showed that signaling from germ cells supports protein turnover in somatic tissues (Ermolaeva et al., Nature, 2013), indicating that ubiquitin-mediated protein turnover could be under cell non-autonomous control. Intriguingly, in addition to germline signals, we found that abrogation of neuronal activity similarly affects protein turnover in somatic cells. Based on genetic screens, we identified central regulators that are important in transducing an as yet unidentified signal from neurons to the soma. Gene expression studies revealed several neuron-specific genes that are differentially regulated and may hence be acting as intercellular communication signals to coordinate protein degradation. Thus, our data suggest that neuronal signaling influences protein turnover in somatic tissues via novel neuroendocrine signaling pathways.
Regulation of Genome Maintenance by Chromatin-Associated Protein Degradation
In recent years, it has become more and more apparent that the ubiquitin/proteasome system (UPS) is central to maintain genome stability and regulate cellular DNA damage response (DDR) mechanisms. Our recent work revealed a central role of the ubiquitin-selective chaperone CDC-48/p97 in regulating the activity and turnover of ubiquitylated proteins bound to chromatin (Dantuma & Hoppe, Trends Cell Biol., 2012). In line with these observations, we uncovered a conserved role of CDC-48/p97 in DNA replication and DSB repair, which is important for cell cycle progression and genome stability (Franz et al., Mol Cell, 2011; Acs et al., Nat Struct Mol Biol., 2011). Similar to loss of replication licensing factors, DNA content is strongly reduced in worms depleted for CDC-48. In addition these worms show hypersensitivity towards replication blocking agents and ionizing radiation, implicating p97 as a potential cancer drug target. Interestingly, CDC-48/p97 interacts with the Werner RecQ helicase, which is involved in DNA replication/repair to repress tumorigenesis and progeria syndromes.
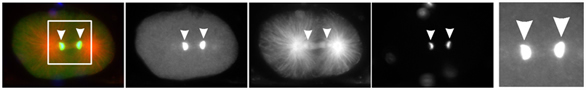
Taken together, we identified the replication licensing factor CDT-1/Cdt1 and the tumor suppressor L3MBTL1 as critical targets of CDC-48/p97. In this context it is interesting to note that p97 is over expressed in diseases, which are linked to rapidly progressive cancer development.
Since the mechanistic role of CDC-48/p97 in DNA metabolism and genome surveillance is unclear, our long-term objective is to define the molecular role of CDC-48/p97 in chromatin-associated protein degradation. To determine the molecular activity of CDC-48/p97 in chromatin associated degradation of the replication licensing factor CDT-1, we combine biochemical approaches with time-lapse DIC microscopy assays in C. elegans embryos To further address mechanistic aspects of chromatin-associated protein turnover by CDC-48/p97, we aim to find the E3 ligase(s), DUBs, and cofactors important for CDT-1 targeting by RNAi-based screens. Given its key role as a ubiquitin-selective segregase, understanding how CDC-48/p97 regulates cell cycle progression and DNA repair would provide novel mechanistic insights into the cross talk between ubiquitin, genome stability, and aging.
Dynamics of Proteostasis Networks in Stress Response and Aging
Cellular differentiation, developmental processes, and environmental factors challenge the integrity of the proteome in every eukaryotic cell. The maintenance of protein homeostasis, or proteostasis, involves the degradation of misfolded and damaged proteins, and is essential for cellular function, organismal growth, and ultimately viability. Sustaining protein quality control (PQC) is not only a long-term challenge for individual cells but also for entire organisms, since damaged proteins accumulate with stress and aging. It is commonly thought that age-related impairment of PQC affects general proteostasis networks, causing enhanced aggregation of misfolded proteins that can be toxic for cells and shortens organismal lifespan. Not all tissues are equally susceptible to the toxicity of protein aggregates, suggesting tissue-specific differences in proteostasis pathways. In humans, aberrant protein aggregation is often associated with neurodegeneration in age-dependent disorders such as Alzheimer’s and Parkinson’s diseases.
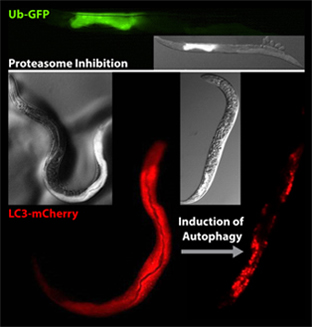
The ubiquitin/proteasome system (UPS) is a major proteolytic route functioning in a cellular network that maintains the proteome during stress and aging. Another proteolytic system supporting protein homeostasis (proteostasis) is the autophagy-lysosome pathway that degrades proteins inside activated autophagosomes. We recently developed in vivo degradation assays based on a fluorescently labelled model substrate that allows monitoring protein turnover in different tissues of C. elegans.
The ubiquitin/proteasome system (UPS) and autophagy are the major proteolytic routes embedded in a cellular quality control network that protects the proteome from long-term damage (Kevei & Hoppe., Nat Struct Mol Biol., 2014). An age-related impairment of either of these systems causes enhanced protein aggregation and contributes to normal aging, suggesting functional overlap and cooperation between UPS and autophagy. Despite searching for key substrates that are destined for degradation, the major challenge in the field is to unravel global (post)transcriptional mechanisms that coordinate proteolytic networks in response to age-related proteotoxicity. We are especially interested in the interplay between UPS and autophagy pathways induced by stress and aging. The identification of central proteolytic pathways and their dynamic regulation will help to understand how protein degradation is fine-tuned in the context of age-related processes. The expected results may contribute to therapeutic intervention against neurodegenerative protein aggregation disorders including Alzheimer’s and Parkinson’s diseases.
Journal Club >>..
Progress Report >>..
Next MitoClub on July 23 >>..
News from the Lab: Elisabeth and Franz Koop Prize for Biochemistry for our Lab member Nitya >>..
News from the Lab: Advanced Postdoc Grant for our Lab member Qiaochu >>..
News from the Lab: Prize for our former Lab member Vishnu >>..
Application call Master fellowships CGA >>..
A Statement against Right-Wing Extremism and for Solidarity >>..